Boug 20 for 1 999 a fare 1. The electric potential energy of interaction between nucleus of an atom and an electron is given by U = 0, where r, is a

PDF) Das Problem in ein Postulat verwandeln: Cassirer und Einsteins Unterscheidung von konstruktiven und Prinzipien-Theorien | Marco Giovanelli - Academia.edu

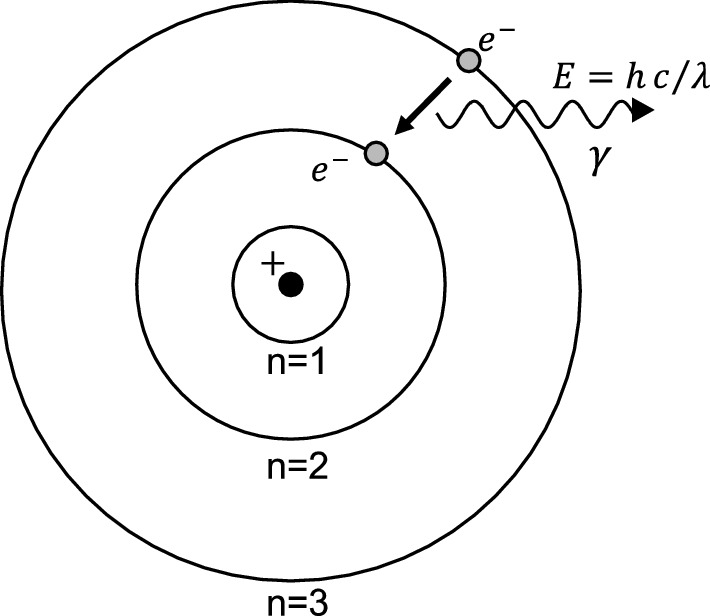
State Bohr's postulate of hydrogen atom which successfully explains the emission lines in the spectrum of

State Bohr's postulate of hydrogen atom which successfully explains the emission lines in the spectrum of hydrogen atom. Use Rydberg formula to determine the wavelength of H, line. [Given : Rydberg constant

Advantages of Bohr’s Theory, Bohr Quantization Postulate, Chemistry Study Material @eMedicalprep.com | eMedicalPrep